
Ask any climate scientist how we should power our world without fossil fuels, and they’re bound to tell you about wind and solar power.
You might be surprised to know that both of these come from the sun. Solar panels collect the sun’s energy directly, but we wouldn’t even have wind if not for the sun.
Why? Because in order to move, you need energy. And not just you. I’m talking about every speck of material on Planet Earth that shifts an inch. It’s because it has energy.
That energy can come from a lot of places. Earth is still a dynamic world with a hot interior, but it’s not hot enough to sustain all the life and other movement on its surface. A lot of our planet’s energy comes from the sun.
But here’s the big question. How the heck does it get here?
Before we dive into that, let’s take a closer look at how the sun generates its energy.
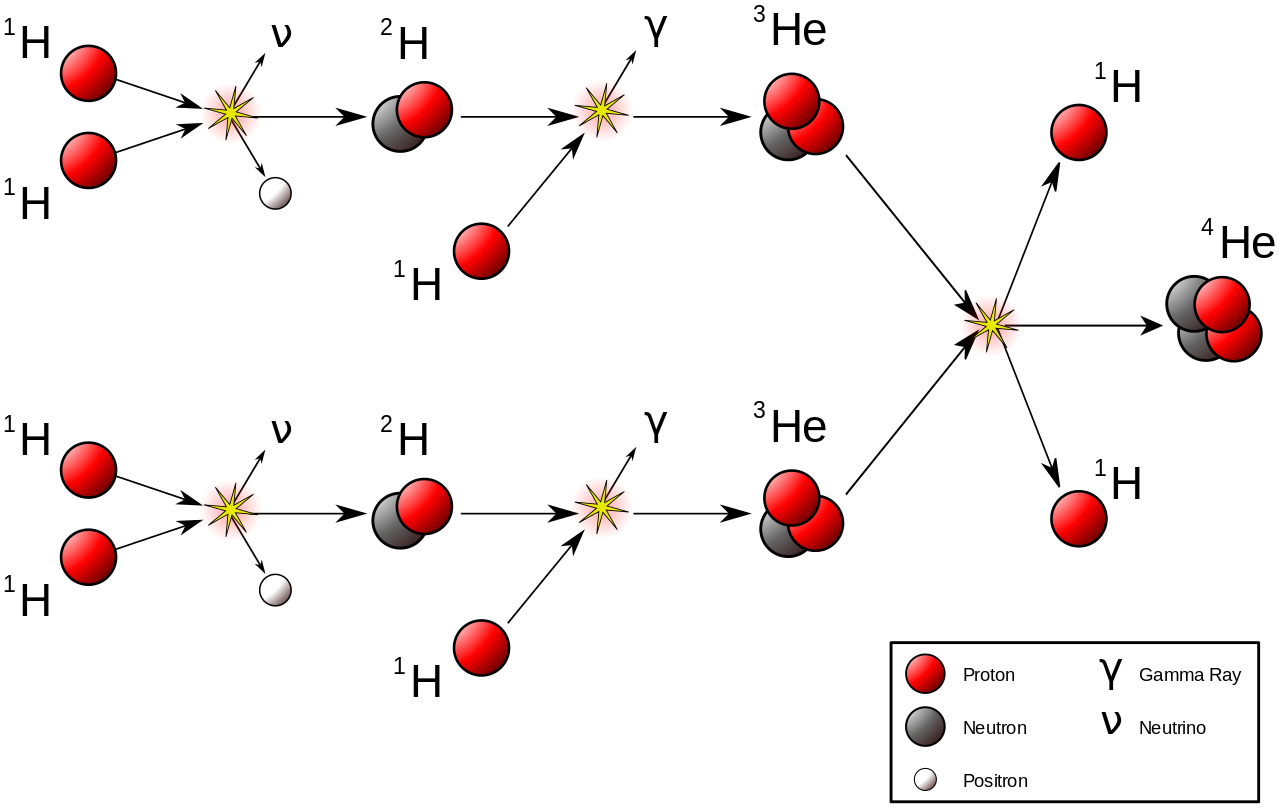
This is a chain reaction called the proton-proton chain. I explained it in depth in my last post. Here, all you need to know is that this chain reaction is basically the battery of the sun. It’s also known as hydrogen fusion.
We know that the sun is extremely powerful.
It generates—and radiates—enough energy to power not only a whole world, but a whole solar system, for billions of years. Theoretically, it’s even possible to harness the sun’s entire energy output and never have to worry about powering our planet.
But the proton-proton chain itself doesn’t actually generate that much energy.
Yes, you read that right.
The energy output of the proton-proton chain is so weak it’s crazy. It can’t even raise a housefly one-thousandth of an inch.
Consider what that means for a second. How easy is it for you to pick up a housefly?
Now imagine not being able to raise it even an inch off the ground, it’s that heavy for you. And it’s worse than that—divide an inch up into a thousand pieces, and you can’t even lift that housefly that tiny distance.
Yeah. Seriously. The proton-proton chain is tremendously pathetic compared to your strength as a human being.
So how come it works so well for the sun?
Well…consider this. How big are you compared to the sun?
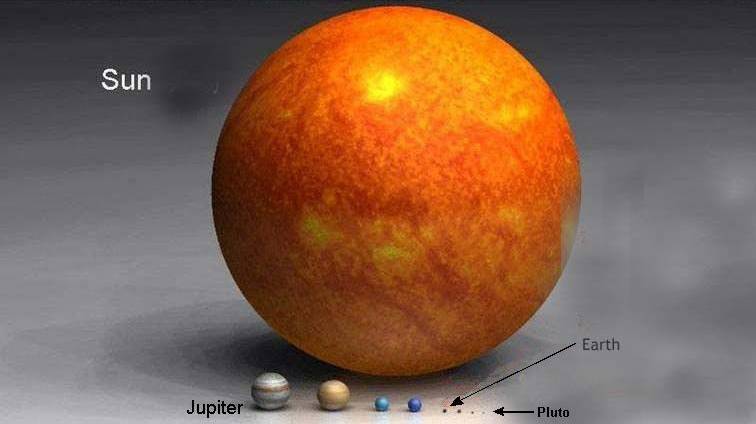
Yeah, that’s what I thought.
About 7.6 billion humans currently fit on the surface of our planet…a planet which is little more than a tiny grain of dust compared to the sun.
If you had a proton-proton chain fusing hydrogen in your body, it wouldn’t work. You really wouldn’t be able to lift a housefly one-thousandth of an inch. But you would still be able to raise it one-millionth of an inch, right?
That means you are generating energy. Not a whole lot, but it’s happening.
In order to generate enough energy to lift that housefly, all you need to do is fuse more hydrogen. A lot more. You need a whole lot more proton-proton chains happening at once.
That’s what’s going on inside the sun, deep within the core. Except the sun’s core contains more matter even than the Earth—about 1,000,000 times more. And the Earth is about 60,000,000,000,000,000,000,000 times the size of a human being.
Suffice to say, there’s enough hydrogen fusion going on to produce a lot of energy. Way more than we need to lift a housefly. Definitely enough to power a solar system.
But we’re still talking about the sun’s core. Yes, it’s powerful enough to power the solar system, but how the heck does it get out there?
When energy from hydrogen fusion starts out in the core of the sun, it’s about 15 million K. That’s close to 15 million degrees Celsius, and 27 million degrees Fahrenheit. But the sun’s surface is only about 5800 K—5527℃, and 9980℉. That’s a huge difference.
That tells us that energy must be radiating out from the sun’s core—and getting a bit lost as it goes, so only a little bit of it remains by the time it gets to the surface.
That’s exactly what happens.
Energy travels out from the sun’s core in the form of a photon, which is a bundle of electromagnetic waves. That’s radiation, by the way. It’s why we call the part of the sun where this happens the radiative zone.
In case you need a refresher on radiation, here’s the electromagnetic spectrum…
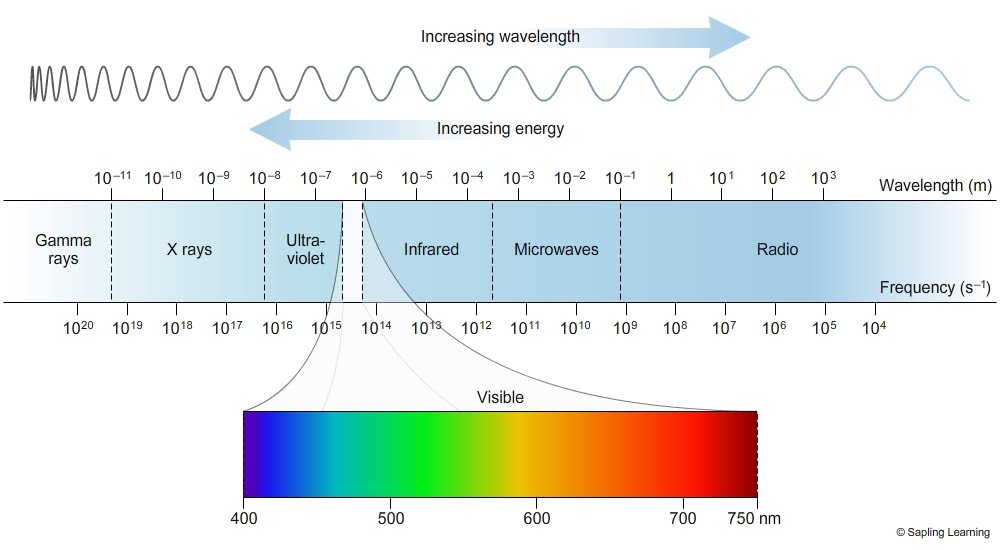
In earlier posts, including this one, I’ve explained how light and energy aren’t all that different. Light as we know it is just one tiny piece of a larger spectrum of radiation, as you can see above.
All radiation has a wavelength, and it gets shorter the more energy the radiation carries. You can see this with gamma rays and radio waves—notice how that wavy line above is very tightly compressed for gamma rays, but much more stretched out for radio waves?
You can think of a single photon within the sun as a messenger, a carrier, for energy from hydrogen fusion. Imagine that the energy is mail that must be delivered to the planets, but a photon has to carry it out of the sun first.
So…what kind of photon are we talking about?
Well, you can probably imagine that with all the hydrogen fusion reactions going on in the sun’s core, it’s a very hot place. An extremely hot place, full of ionized gases. So we’ll really only find one kind of photon in there, and it’s the one with the most energy.
Take a look at the electromagnetic spectrum up above. Which one do you think it is?
If you said it’s a gamma ray, you’re right. And that gamma ray is going to carry the sun’s energy far out from the core, until it starts traveling through cooler and cooler gases. And eventually, the gas will absorb the photon.
Wait a minute. Isn’t that photon supposed to reach the solar system somehow?
You’re right—it doesn’t disappear. It’s going to get reemitted as two X-rays.
And those two X-rays don’t survive forever, either. They’ll eventually get absorbed and reemitted, and by the time our energy packet reaches the edge of the radiative zone, it’ll be carried by a bunch of longer-wavelength photons.
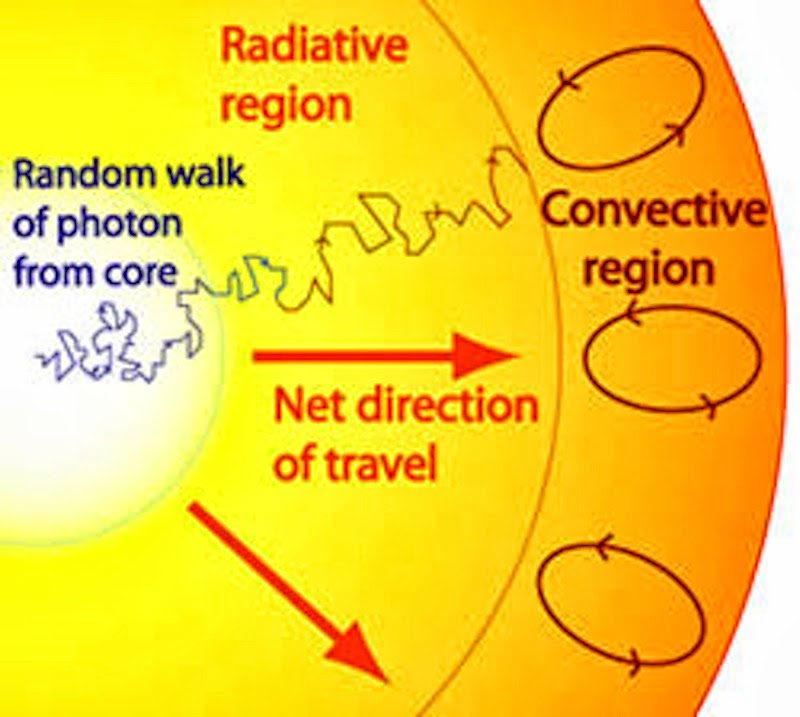
Now, our energy packet has reached the convective zone.
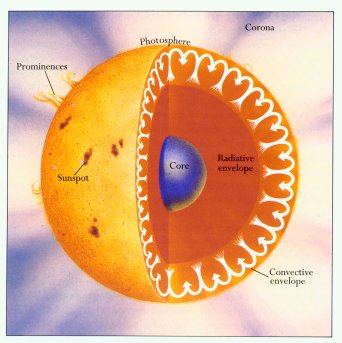
I really like this image because it shows convection in a way a lot of diagrams don’t. Usually, it’s portrayed as a bunch of ovals with arrows. And that’s the simplest way to show it, but it’s not entirely accurate.
This diagram shows you what really happens.
As our photons reach the edge of the radiative zone, they are greeted by layers of much cooler gas. Of course, it’s not cold by any means, but it’s very cool by the sun’s standards. It’s too cool for the photons to pass through. So it’s like being stuck behind a dam.
These photons won’t give up easily, though. They still want to radiate outward—but they can’t. So they keep trying to push at the convective zone’s dam, and the gases there start to heat up.
Have you ever heard the phrase, “Heat rises”?
When we say that heat rises, we mean that it escapes away from the nearest gravity source. But the other part of the equation that we don’t always remember is that when heat rises, cold sinks.
That’s what’s happening in the convective zone. Those hot gases in the inner layers rise up, as shown by the paths of the arrows in the diagram. But as they get farther from the core, they cool off, and they start to sink back down.
Energy can stay stuck in the convective zone for millions of years. But eventually, it will make its way to the sun’s surface—the photosphere. And when it finally gets there, it’ll radiate out into space as about 1800 photons of visible light.
So there you have it: the reason why the sun’s core supports a whole solar system. Energy does escape. And visible light is no different from any other form of radiation in that it has energy. Enough to power a world.
But here’s the real question. How do we know that any of this is true?
Well, I’m glad you asked! We’ll cover the answer in my next post.